One of the main reasons pelvic organ prolapse (POP) occurs is due to childbirth. The stress of birth can, in some cases, shift the organs in the pelvis from their normal positions. The most common organs affected during childbirth are the bladder and the uterus itself. When this occurs, the symptoms can be mild or severe, depending on the type and extent of the prolapse.
The most common symptom is a feeling of fullness or pressure in the lower abdomen. Frequent urination and incontinence are common with a prolapsed bladder. As the prolapse worsens, the feeling of pressure can change to pain, and the condition can be very uncomfortable and debilitating.
There are several treatments for pelvic organ prolapse, and recently, the use of an implant called transvaginal mesh has become very common. During this procedure, a light mesh material is inserted through the vagina to help secure the prolapsed organs. While in theory this seems like a great solution for pelvic organ prolapse, in reality there have been numerous complications associated with this procedure.
These complications have been reported with increasing frequency to the Food and Drug Administration (FDA). Between the years of 2008 and 2010, more than 2,800 complaints about the transvaginal mesh procedure were logged. The FDA has made these complaints public in order to help patients realize the risk they are facing if they elect to go ahead with this procedure.
The most common side effect following transvaginal mesh surgery is the erosion of the mesh into other organs. This erosion can cause bleeding and other side effects. Another common complication is the actual wearing through of the vaginal walls, as the mesh presses against them. This is a very serious complication that can result in internal bleeding and serious infections. Because of such severe complications, mesh users have started to file a transvaginal mesh lawsuit against the mesh manufacturers.
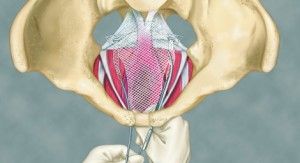
Other side effects noted by the FDA include pain and neuromuscular problems following the procedure. Unfortunately, many doctors are not aware of the complications that their patients are facing with this procedure. It appears that few are properly trained in the insertion of the transvaginal mesh, and this in part explains the high rate of complications associated with the procedure.
The FDA is continuing to monitor the problems associated with transvaginal mesh surgery. The agency has ordered dozens of mesh manufacturers to conduct safety studies on their products.
If your doctor has recommended transvaginal mesh, it is vital to understand the potential risks you may face. While all surgical procedures carry risks, the number of complaints associated with mesh are cause for real concern.
Ask your doctor if there are alternatives to this procedure. For minor cases, simple muscle exercises can be used to help shift the prolapsed organs back into place and strengthen the vaginal walls. Serious cases can be fixed with a surgical procedure that does not include the insertion of the mesh.
If you feel that you are not receiving enough treatment options, don’t be afraid to ask for a second opinion from another surgeon or specialist. You deserve to know the true complications associated with transvaginal mesh, and you deserve to have a surgeon who has been properly trained in the procedure before going ahead with surgery.

The mesh surgery comes with many risks, including the fact that the mesh goes into your body pliable, but hardens over the years and turns into something the consistency of a large screen, like metal. Women complain of feeling the mesh poking them when they move in certain ways. The mesh can even penegrate the walls of the vagina causing discomfort to a woman and her sexual partners, who can feel the mesh poking them during intercourse. Please consider physical therapy for the treatment of prolapse and incontinence. I actually guest host a radio show here in Houston and we will be discussing incontinence on the show on Monday.
ReplyDeleteIncontinence. The very mention of the condition brings feelings of shame to its sufferers and embarassment to those who are free from its effects. Incidence, for this reason, is extremely under-reported, but even these inaccurate statstics tell us that 1 in 10 of us experience incontinence that soaks our clothes at some point in our lives. So if you've ever sneezed, laughed or coughed and leaked or if you know where the bathrooms are everywhere you go and you are constantly making a bee-line for one, join me Monday when my guest on Whole Mother will be Dr. Michelle Howze, expert in pelvic floor physical therapy, who will teach us simple techniques to make it better.
Whole Mother, Monday, July 30, live 6:30 - 7 a.m.
... 90.1 FM in Houston, Texas ; 89.5 in Galveston, Texas; 89.7 in Huntsville, 90.3 in Livingston;
Archived minutes after the live broadcast at KPFT.org (click on "On Demand" and scroll down to Monday's show)
See you on the radio,
Debbie Hull
Several breakthroughs would be discovered in the future so that this problem would have a working solution. Thanks to technology and the undying curiosity of humans, we can safely say, that there will be a time when they will find a perfect solution to this. For the moment, we just have to make do with the solutions that are presented to us.
ReplyDeleteMy grandmother was diagnosed a while ago with a prolapse. She was in agony all day long.She wasn't up to going through surgery, so we were searching for something/anything to relieve her pain. A good friend of mine suggested I contact Fembrace to see if they can help. We went online & got their info and we ordered the V-Brace. My grandmother and I will forever be grateful to my friend for making the suggestion. I took upon myself to spread the word as much as I can
ReplyDeleteTheir contact info is:
Web www.fembrace.com
Email info@fembrace.com
Phone (877) 535-6800
Best of health to all!
I used the V-Brace in pregnency.It gives the support when you feel the pressure of the baby pushing down. I also got it from Fembrace....
DeleteI feel sorry for all those who endured pregnancy for many months and then wake up with yet another kind of illness. I just wish they can find people who will understand and comfort them in these difficult times of their lives. It's a shame that such a product expected to bring relief actually did more harm than good. What I'm talking about are the vaginal mesh implants.
ReplyDelete